Frey’s Syndrome / Gustatory Sweating
It is also known as Gustatory Hyper-Hydrosis, Auriculo-Temporal Syndrome or Baillarger Syndrome.
Definition:
Gustatory (Taste or the technical term, gustation; adj: gustatory) sweating was first described by Duphenix in 1757 in association with parotid gland trauma, then by Baillarger in 1853 and subsequently by Łucja Frey in 1923; not only in association with parotid gland trauma but confined to the distribution of the auriculo-temporal nerve.
Frey’s Syndrome (FS) is the phenomenon secondary to gustatory stimulus, manifested by flushing and sweating of parotid, frontal and sub-mandibular areas consequent to trauma to the parotid or another major salivary glands (ie sub-mandibular or sub-lingual) often either after a parotidectomy [link to Superficial Parotidectomy webpage] or excision of the sub-mandibular gland [link to Submandibular Gland Removal webpage].
In other words, FS is characterised by excessive sweating of the forehead, upper lip, peri-oral region or sternum subsequent to gustatory stimuli. Other regions can be affected in FS.
These include the:
- Occipital region
- Cervical region
- Ear region
- Temporal hair region
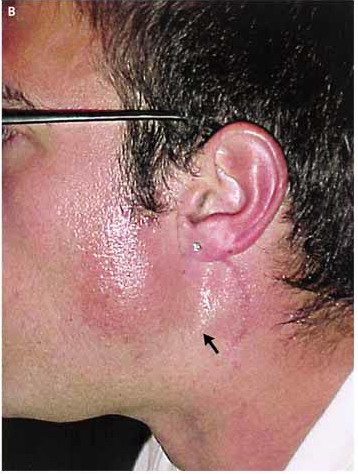
Photo showing Gustatory Sweating (arrowed). The Superficial Parotidectomy [link to Superficial Parotidectomy webpage] scar can be seen.
The Auriculo-Temporal Syndrome features facial flushing or sweating limited to the distribution of the Auriculo-Temporal Nerve [files/Auriculo-Temporal_Nerve.jpg] and may develop after trauma to the Parotid Gland, in association with parotid neoplasms (benign / malignant cancers) or following their surgical removal [link to Superficial Parotidectomy webpage].
FS is more commonly seen after:
- Penetrating wound of the parotid region
- Chronic infection of the parotid region
- Parotitis
- Parotid surgery [link to Superficial Parotidectomy webpage]
- Jugulo-carotid lymph node dissection
- Submandibular gland surgery [link to Submandibular Gland Removal webpage]
- Carotid endarterectomy
- Trauma of the Auriculo-Temporal Nerve after forceps delivery
- Mandibular [link to Fractured Mandible webpage] & zygomatic [link to Fractured Zygoma webpage] fractures
- TMJ surgery
- Internal maxillary artery pseudo-aneurysm
- Chorda tympani injury
- Cervico-thoracic sympathectomy
- Tumour infiltration of the upper node of the sympathetic plexus
- Diabetes mellitus
- Herpes zoster
- Platinum-induced neuropathy
- CNS diseases (syringomyelia, CVA / stroke, encephalitis)
- Loss of insulation twixt the post-ganglionic sympathetic & para-sympathetic nerve sheaths within the Auriculo-Temporal Nerve
FS is an unusual complication after Sub-Mandibular Salivary Gland Excision [link to Submandibular Gland Removal webpage] and neck dissections. When reported in relation to neck dissection, it has been associated with ‘radical’ neck dissections, particularly for thyroid cancer and has tended to occur in the sub-mandibular area on the upper skin flap.
Incidence:
FS has been reported in > 60% of patients but is usually symptomatic in < 10% who seek treatment.
Pathophysiology:
FS is related to aberrant regeneration of nerve fibres. Hence, sweating or dermal flush occurs during salivary stimulation (ie eating or thinking of eating / food).
Parotid Gland. FS is believed to be the result of mis-directed autonomic nerve regeneration following injury to the parotid region. After injury, the sectioned post-ganglionic secreto-motor para-sympathetic fibres (Auriculo-Temporal Nerves) which normally innervate the Parotid Gland, become connected to sympathetic receptors which innervate sweat glands. Hence, stimuli that normally cause salivation (aromatic foods, thinking about certain foods) simultaneously cause pathologic sweating and flushing in the pre-auricular area on the side of the nerve injury.
Submandibular Gland. The innervation of the sub-mandibular gland is similar to that of the parotid gland; the difference involves pre-ganglionic para-sympathetic fibres that originate in the Superior Salivatory Nucleus and travel along the Facial Nerve and the Chorda Tympani to the sub-mandibular ganglion. Post-ganglionic para-sympathetic fibres originate in the sub-mandibular ganglion and travel to the sub-mandibular gland. Pre-ganglionic sympathetic fibres originate in the 1st & 2nd Thoracic Spinal Nerves, synapse in the Superior Cervical Ganglion with post-ganglionic sympathetic fibres and travel along the External Carotid Artery and the Facial Artery to the sweat glands.
During surgical excision of the Sub-Mandibular Gland, aberrant post-ganglionic para-sympathetic fibres can subsequently innervate severed post-ganglionic sympathetic fibres, supplying the sweat glands in a misdirected pattern. This results in a hyperhidrosis of the affected area ± concomitant erythema (reddening) caused by parasympathetic vascular effects at the site.
Other nerves might be involved such as the Facial Nerve, the anterior & posterior branches of the Greater Auricular Nerve & the Lesser Occipital nerves served as guiding structures for the regenerating parasympathetic nerve fibres. Although FS does not cause significant physiological harm, profuse gustatory flushing and sweating can cause social and psychological distress in some patients.

Diagnosis:
FS / Hyperhidrosis is diagnosed by history and examination but can be confirmed with the iodine and starch test (apply iodine solution to the affected area, let dry, dust on corn starch: areas of sweating appear dark).
Testing is necessary only to confirm foci of sweating (as in FS or to locate the area needing surgical or botulinum toxin treatment) or in a semi-quantitative way when following the course of treatment.
Minor Starch-Iodine Test
This test entails ‘painting’ the affected side of the face with iodine and waiting for it to dry. The area is subsequently powdered with cornstarch which renders visible the sweating reaction. To elicit salivation / sweating, the patient is then asked to chew a lemon slice for 5 minutes. The appearance of black spots over the starched field constitutes a positive result, generated by a chemical reaction between iodine, dissolved starch and sweat, confirming sudoresis (sweating) secondary to gustation.
The margins of the black spots are drawn with a ball-point pen.
As alluded to earlier, the black spots can be used to pinpoint where to use the Botox and gives a semi-quantitative recording of how the gustatory sweating is improving (or not).
The Minor Starch-Iodine Test has drawbacks:
- difficult to use in hair-bearing areas of the skin
- does not allow evaluation of the severity of the gustatory sweating
Other methods have been tried but have not gained widespread usage.
- Weighing filter paper
- Thin facial tissue papers
- Iodine Paper Histogram
- 1 step methods using dyes
- Impression materials using silicone or polyvinyl
- Bio-sensoring methods with enzymatic electrodes
- Infra-red medical thermography
- Evaluation of evaporation
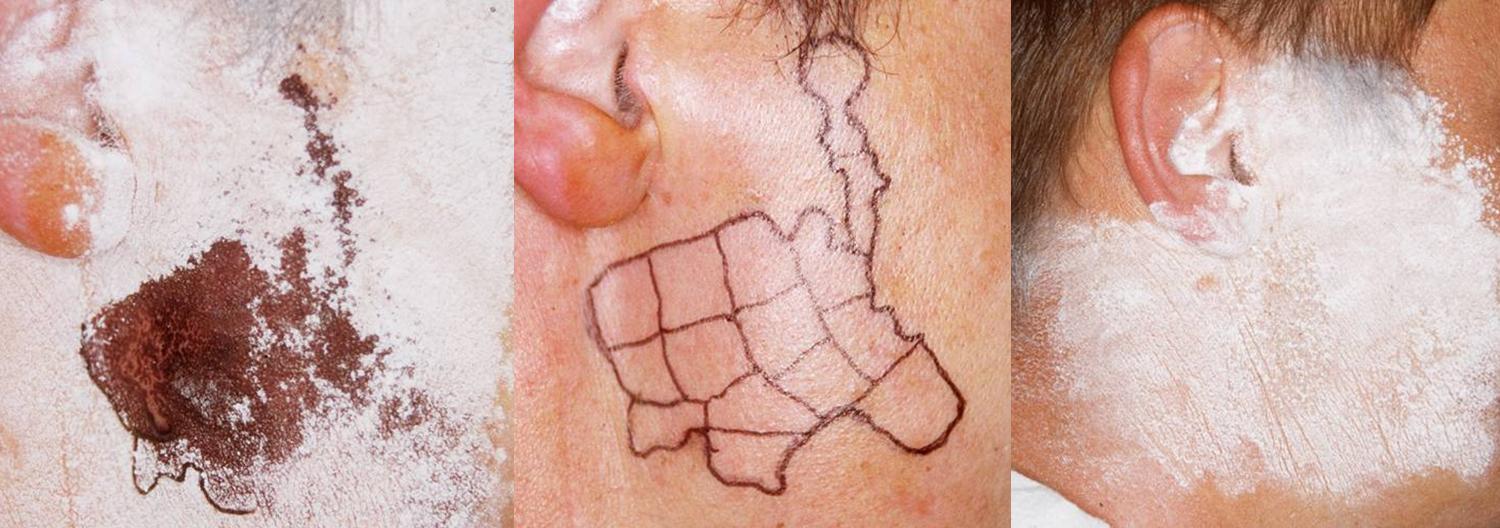
Photos showing the Minor Starch-Iodine Test & the Use (& Efficacy) of Botox to Ameliorate FS
Treatment:
In most cases, FS patients do not complain of their symptoms and are often treated effectively with topical anti-perspirant gels applied to the affected area.
However, when symptoms become bothersome, various prophylactic and therapeutic surgical strategies have been proposed to minimise the incidence or severity of FS following Parotidectomy [link to Superficial Parotidectomy webpage].
These include:
- Botox (patients who are unresponsive to topical therapy may want to consider a trial of botox before considering surgical options)
- Topical anti-perspirant (20% aluminium chloride solution)
- Application of an ointment containing an anti-cholinergic drug such as 3% scopolamine, 2% glycopyrolate or diphemanil methylsulphate
- Topical application of a adrenoceptor agonist (clonidine)
- Blockage of parasympathetic outflow by way of alcohol injection or 2% lignocaine injections at various sites such as the Otic Ganglion & the Auriculo-Temporal Nerve
None of these approaches allows a definitive cure and relief is only temporary. Botox appears to be the easiest and safest method that provides the longest period of relief with the lowest morbidity & adverse incidents rates.
- Radiation to the affected skin region causes skin atrophy and is a highly efficient methods in patients with FS. However, this option is not used anymore because of the high risk of radiation-induced skin carcinoma.
- Temporal Fascia grafting
- Application of synthetic materials to the surgical field at the time of surgery
- Ligature (transaction / resection) of the Auriculo-Temporal and Chorda Tympani Nerves.
- Tympanic Neurectomy
- Intra-Cranial division of the 9th Cranial Nerve (Glossopharyngeal Nerve).
However, none of these surgical procedures results in definitive cures because anastomotic connections between the Greater & Lesser Superficial Petrosal Nerves allow aberrant regeneration pathways and none is without significant risk of major morbidity.

Useful Websites:
Useful Articles:
Clin Auton Res 2002. Editorial. Mechanism of gustatory flushing in Frey’s syndrome.
Arch Facial Plast Surg 2003. Use of AlloDerm Implant to Prevent Frey Syndrome After Parotidectomy.
Arq Neuropsiquiatr 2003. Botulinum Toxin for Treatment of Frey’s Syndrome. Report of 2 cases.
JADC 2009. Management of Frey Syndrome Using Botulinum Neurotoxin – A Case Report.
The Open Dermatology Journal 2009. Hyperhidrosis – A Review of a Medical Condition.
BJOMS 2009. Short communication. An unusual cause of Frey syndrome.
Cochrane Collaboration 2012. Interventions for the Treatment of Frey’s Syndrome (Protocol).